Archaeological evidence indicates that people have been using Silver for over 5000 years, Silver being one of the famous Metals of Antiquity, mainly used for manufacturing jewellery, silverware and coinage.
Even today, Silver is very much used for necklaces, cuff links, bracelets, body jewellery and belt buckles, sterling silver containing about 92.5% silver and 7.5% copper.
Since the 20th century, Silver and its compounds have also been used in electrical conductors, electric contacts, catalyst supports, appliances, lenses, chemical machinery, photography, and brazing alloys, as a provider of filtering devices for drinking water, products of healthcare and medical instruments.
Regarding its general properties, Silver (Ag) is a white lustrous metal, well known for its electrical conductivity. It also displays a very high thermal conductivity, as well as reflectivity.
Silver ores, such as horn silver (AgCl) and argentite (Ag2S), are used for mining the pure silver and in combination with deposits of ores containing lead, gold, or copper.
Silver belongs to Period 5 and Group 11 (Ib) of the periodic table. Located between Copper (Period 4) and Gold (Period 6) in the periodic table, its chemical properties lie between these metals. Silver has an atomic number of 47 and the atomic symbol of silver is Ag (Ag is from the Latin word: Argentum, and where Argentium Silver derives its name from).
Silver frequently plays second fiddle to some other precious metals like gold, but it has unique properties of its own, that deserve a good look.
Important Advantages of using Silver
- Silver is soft, malleable (easily squashed) and ductile (easily stretched).
- Silver is highly reflective.
- Silver can be polished to a luminous sheen.
- It is inexpensive compared to gold and platinum.
- Silver has good antibacterial properties for items like cutlery.
- Silver is durable and has a longer lifespan compared to other metals and costume jewellery.
For a detailed description of Silver as a material, the physical, chemical, mechanical and thermal properties can provide an insight:
Physical Properties of Silver
- Silver is a white, dense, and relatively soft metal.
- Silver can be polished to a high degree.
- Excellent electrical and heat conductor.
- It is highly malleable. It can be easily shaped or bent.
- It has the ability to be dissolved.
- Silver has a ‘Solid’ phase at room temperature.
- Silver has an Atomic weight of 107.87 g.mol-1
- Its Density is 10.501 grams per cubic centimeter at 20oC (68 F).
- The number of isotopes present in silver is 66. Out of 66, only 2 are stable isotopes.
- The most common isotopes are Ag-107 and Ag-109. Ag-107 has 51.839 % natural abundance while Ag-109 has 48.161 % natural abundance.
Chemical Properties of Silver
- At normal temperature, it does not react with O2, N2, or H2.
- Most of the complex compounds of which silver form are soluble in water.
- Silver requires cyanidation to be extracted from the ores. Cyanidation of silver is done by its dissolution in an alkaline sodium cyanide solution along with an inlet for air.
- The melting point of Silver is 1,763.2 F, while the Boiling point is 3,924 F.
- It has an Electronegativity of 1.93 and Electrode potential of 0.799 V.
- The Electrochemical equivalent of Silver is 4.025 g/A/h.

Mechanical Properties of Silver
- According to the Mohs Hardness Scale, Silver is 2.5-3, meaning pure silver is a soft material.
- The tensile strength of Silver is 140 MPa.
- Its Poisson’s ratio is 0.37.
- Its Modulus of elasticity stands at 76 GPa.
- The Silver has a Shear modulus of 27.8 GPa.
Thermal Properties of Silver
- The Thermal expansion coefficient of Silver at 20-100°C/68-212°F is 19.6 µm/m°C .
- Its Thermal conductivity is 419 W/mK.
..............................................................
The white metal, Silver, has an eminent prominence for its use in different markets all across the globe, from electronics, coins, cell phones, solar panels and to the jewellery market.
Silver jewellery has a huge global fan base and it fits well into many fashion styles. A metal that is priced on the lower side, there are also more design possibilities to be had when making jewellery in silver.
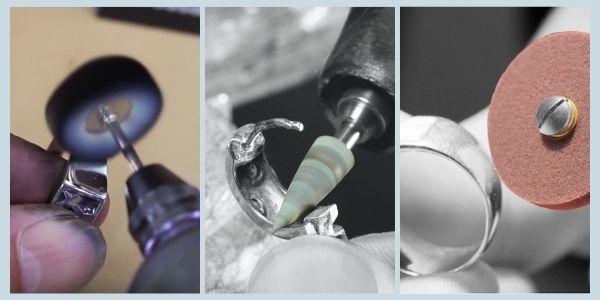
10 Silver Jewellery Making Processes
In it's crudest form Silver metal sheeting can be pressed into dies to form concave shapes ready to be hammered or soldered. It can be forced through a hole, referred to as 'drawing', and extruded as wire or a piece of tubing, this can then be used to make rings, bangles, and chain etc.

1. Casting (Lost Wax Casting)
This is the process whereby wax designs are surrounded by plaster to create a mold. Molten metal is then poured into the mold. After the piece is cooled down, it is cleaned of the residue with a white silicone wheel abrasive, polished and finished.
2. Electroforming
This is a more complex process, by which Silver is electroplated onto a shape, commonly made of wax. This shape is then introduced into a bath, treated with a special solution to form thicker silver layers on top of the base-metal layer. Later, the shape is warmed up, the wax from the inside melts and, a finished piece of silver will remain.
3. Cold Enameling
This is a manufacturing process based on the use of cold enamel which is a type of epoxy resin. This resin is applied to metal like a paint, then the piece is heated, bonding the paint with the silver.
4. Gold Sheeting
Another way to manipulate silver, is based on the process of laminating a sheet of gold onto a sheet of sterling silver. In the end, from the final product, the shapes are cut and assembled into jewellery.
5. Stamping
A common process to make light-weight jewellery. Dies, resembling cookie cutters, are used to stamp silver sheets in order to form cut-out shapes which will be later put together to form jewellery. Many jewellery items are made in this way, such as pendants, earrings, and charms.
6. Oxidisation
To give silver jewellery an antique appearance, oxidation is often used as a process, by which silver is treated with a solution to make it darker. Later, the silver piece is polished and the black areas remain in the indentations. A nice finish, but one that will eventually wear off and will need to be applied again at a later date.
7. Plating
Another method used to manipulate Silver is the plating process, the most common being gold, rhodium, black rhodium and anti-tarnish plating. The plating of silver jewellery is made by soaking the jewellery in a solution, passing an electric current through it, which then leads to the formation of a thin layer building up on the jewellery. The purpose of the plating process is to increase the scratch resistance and to improve the appearance.
8. Sawing, cutting and drilling
Silver can be cut into shape using steel saw blades in a jewellers piercing saw frame, and drilled using HSS (high speed steel) drill bits in a rotary tool. Read more here on how to use a jewellery saw and saw blades to cut and shape your silver.

9. Filing, sanding and texturing
Once the shapes have been cut out as mentioned above, the silver can be filed with steel files to remove any snags, or sharp and rough edges. Sand down through various grits before pre-polishing or polishing, or texturing to produce a satin or frosted finish by using a variety of hand polishers or rotary tool accessories.
To find out more about the correct files to use for jewellery making and how to use them here.

10. Smoothing, pre-polishing and polishing
Silver can be left with a textured surface to your desired effect, or be smoothed, pre-polished and polished to a high lustre finish by using a variety of hand polishers, compounds, or rotary tool polishing accessories. Achieving a great finish can add a high value to silver jewellery.
Medical Applications of Silver
Silver or Silver nanoparticles have been used for a long time to protect against a wide range of gram-negative and gram-positive bacteria, indicating that Silver is toxic to bacteria. Unlike any other antibacterial medicines, Silver doesn’t create resistance or immunity in the organisms that are destroyed by it. Overall, Silver has been a major therapeutic mediator in medicine, particularly in contagious diseases which also includes surgical infections. Its risk to benefit ratio is profitable.
- Silver can be used as an antibiotic coating on medical devices. It has been added to some bandages for its antimicrobial effect.
- Traditionally, Silver nitrate drops were placed into newborn babies’ eyes for many years to shield against blindness caused by maternal gonorrhoea.
- Silver has also been used in salves for burn victims, and to purify water.
- Nanocrystalline Ag+ is used in preventing infection in large body surface area burns.
- Recently, Silver alloy‐coated Foley catheters are used for the avoidance of urinary tract infection in patients.
- The inorganic silver can fight and kill antibiotic-resistant super-pathogens.
- Silver destroys Pseudomonas aeruginosa infections.
Electronic Applications of Silver
- Silver is used in electrical switches as contacts by applying very small quantities.
- Light-emitting diodes (LED) use silver electrodes to produce low-level, energy-efficient light.
- DVDs and CDs can have a skinny silver recording layer.
- Silver is used in batteries that employ silver oxide or silver zinc alloys.
- Silver, when paired with any superconductor, can transmit electricity even more efficiently than the superconductor unaided.
- Nanosilver conductive inks help in Printed Electronics.
Industrial Applications of Silver
- Silver acts as a catalyst to produce ethylene oxide and formaldehyde.
- Silver catalysts can also be used to yield formaldehyde which is the important element of solid plastics adhesives.
- Mirrors can be made by placing silver on glass or metals by the process of chemical deposition or electro-deposition.
However, where silver has its benefits, it also has a fair share of drawbacks...
Drawbacks of Silver
- Silver discolours when exposed to sulfur-containing gases in the air. Silver jewellery is oftentimes unpopular due to this reason.
- Frequent and lengthy interaction with skin may cause allergic dermatitis.
- Exposure to high concentrations of vapours may cause respiratory irritation.
- Soluble silver salts like AgNO3, are deadly in concentrations of up to 2g.
- Silver compounds can be gradually absorbed by body tissues, resulting in bluish or blackish skin pigmentation.
- Silver may cause severe corneal injury if liquid comes in contact with the eyes during an experiment.
- If swallowed, it can cause stomach discomfort, nausea, vomiting, diarrhea, and narcosis.
- Silver can cause permanent brain and nervous system damage as a consequence of repetitive exposure.
- Chronic overexposure to a component or components in silver have been found to cause organ damage in laboratory animals.
Target Markets of Silver
As we can see from the benefits and drawbacks of silver, even though it requires careful handling in every application, it also covers a wide range of markets that have been using silver as an essential element for a long time.
Domestic uses estimated in 2019 for Silver were: 30% electronics; 26% jewellery and silverware; 12% coins and medals; 3% arts, and 29% others. As of 2020, more than half of the world's Silver is used in industrial applications.
Other uses for silver include the use of antimicrobial, clothes, medicinal and plastic bandages, batteries, silver covers/ coatings, brazing and soldering, autocatalytic converters, galvanizing, inks, mirrors, photovoltaic solar cells, water purification, and wood processing.
The photographic silver demand has dropped as digital images have become popular, plus jewellery and electrical devices have also shown signs of deflation. But the demand for Brazing alloys, coins, silverware, etc. seems to be increasing. Overall, the world demand for silver is expected to rise.

Silver Jewellery Market in the UK
Precious metals like Gold and Silver are the largest segment in the UK jewellery market. The total Market Size is currently £4.75bn.
Proper procedures and regulations are in place to certify the metals. Hallmarking is an ancient law that has been in the UK since the 1300’s to ratify valuable metals.
According to the UK Jewelry Market, 2017-2022 report, it is estimated that the jewellery market in the UK will grow in the next five years by £687m.
Silver Jewellery Market in the USA
The United States is marked as the world’s number one jewellery market. The US jewellery market has experienced a path of smooth growth in the past few years and market is expected to grow even further from 2020 to 2024.
The US jewellery market and especially the silver market is driven by various growth-enhancing factors such as rising per capita disposable income, a growing number of digital buyers, soaring urban population and an increase in the female population.
The United States Geological Survey (USGS) has tabulated the amount of silver used in the United States by category of use. According to the survey, 6% of the silver is used for jewellery and silverware.
In 2018, the value of the jewellery market in the United States amounted to approximately 36.77bn USD. This market was forecast to reach a value of over 60bn U.S. dollars by 2025.
Silver Jewellery Market in Europe
Being the second largest jewellery market after the USA, the EU represents 20% of the world jewellery market. EU consumers spent € 23,955mn, with Italy, UK, France and Germany making up to 70%.
Silver jewellery is moving up in the number of sales and is benefiting from high gold prices.
Silver is second only to gold in terms of numbers of items sold, not only in Italy, Germany, Spain and Scandinavian countries, but also in the Eastern EU states.
In terms of value, EU silver jewellery retail sales in 2015-2018 were estimated at €4,311M. Regarding the volume consumption, the silver jewellery market has increased overall in the European countries.
Silver Mining
Regarding the extraction of Silver, it cannot be mined in bulk due to the comparative costs with Gold.
The mining and polishing of one ounce of gold takes $350-$450 to mine, refine, and bring to the global market. This is a profitable procedure as gold is being traded at the price of $1200 in the global jewellery market.
Silver is mined primarily as a by-product and the cost to mine and refine it is similar to the cost of mining one ounce of gold and yet Silver is traded at a price of $17-$18 in the global market.
Currently, with silver trading at $17-$18 and gold trading at $1200, it would potentially take 65-70 ounces of silver to buy one ounce of gold.
Only 27% of silver comes from mining where silver is the primary source of revenue. The remaining 73% come where silver is a by-product of other metals like copper, lead, zinc etc.
Silver is mined through a drilling process either by the open pit method or underground methods. The open pit method implies using heavy machinery to mine deposits which are near to the surface. The cost of using this mechanical machinery is much higher than the product being extracted.
In the case of Underground mining, deep shafts are dug into the ground to extract the ore, which will be crushed, grounded and separated through a process of flotation, achieving high silver concentrations. This method is highly profitable.
Altogether, Mexico, China, and Peru account for almost half of the global output of Silver mines.
-----------------------------------------
The white metal has been used for millennia and will continue to be used, the jewellery market being one of its biggest. Besides its mercantile role, silver has had an enduring role in our culture and will continue to do so.


